Synonyms:Benzene,2,4-dichloro-1-methyl-;Toluene, 2,4-dichloro-;2,4-dichloro-1-methylbenzene;2,4-DCT;2,4-DICHLOROTOLUENE;2,4-DICHLOROMETHYLBENZENE;1,3-DICHLORO-4-METHYLBENZENE;2,4-dichloro-1-methyl-benzen
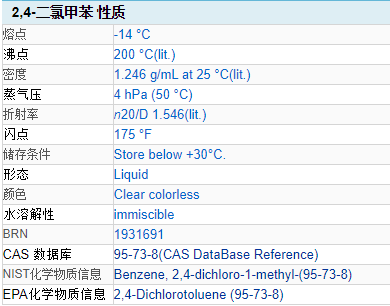
CAS Number: 95-73-8
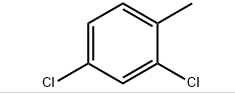
Molecular formula: C7H6Cl2
Molecular weight: 161.03
EINECS number: 202-445-8
Related categories: agricultural and environmental standard products; triazole fungicides; fungicide intermediates; pesticide intermediates; organic raw materials; intermediates; organic intermediates; Organics; Aryl; C7; Halogenated Hydrocarbons; Aromatics; Building Blocks; Chemicalbook Chemical Synthesis; Hydrogenated Hydrocarbons; OrganicBuildingBlocks;Analytical standard products;Organic building blocks;Pesticidesintermediate;Halogenated hydrocarbons;Organic chemical raw materials.
2,4-Dichlorotoluene use and synthesis method
Chemical properties: colorless and transparent liquid.
Use:
1) Used as intermediates of pesticides, dyes and pharmaceuticals, used in the production of 2,4-dichlorobenzaldehyde, drugs such as adipine, buprofen, etc.
2) 2,4-Dichlorotoluene is an intermediate of the bactericides diniconazole and benzylchlorotriazole, and Chemicalbook is also the raw material for the preparation of 2,4-dichlorobenzaldehyde.
3) Organic synthetic raw materials, used in the pharmaceutical industry for the production of antimalarial drugs, Adepine, and the synthesis of ventral acid. Used in pesticide intermediates to manufacture 2,4-dichlorobenzyl chloride, 2,4-dichlorobenzoyl chloride, and 2,4-dichlorobenzoic acid.
There are two synthetic methods for production methods.
1. The 1.2,4-dichlorotoluene method uses 2,4-diaminotoluene as raw material, and is obtained by diazotization and chlorination. Put hydrochloric acid and water into the reaction pot, heat to 50℃, dissolve 2,4-diaminotoluene under stirring, then put hydrochloric acid and cuprous chloride into the pot, add 1% sodium nitrite solution evenly into Chemicalbook , The temperature is maintained at about 60 ℃, stand still for layering, the lower crude product is washed with water until it is neutral, alkali is added to alkali, and then washed with water to remove the alkali, the crude 2,4-dichlorotoluene is separated, and the finished product is steam distilled. . The 2.3-chloro-4-toluidine method is obtained by diazotization reaction with sodium nitrite, and Sandmeyer reaction with copper chloride.
2.The production method and its preparation methods are as follows. In the parachlorotoluene method, p-chlorotoluene and catalyst ZrCl4 are put into the reactor, and chlorine gas is vented to carry out the chlorination reaction. The amount of chlorine gas is controlled to the end of the reaction and the reaction is stopped. The obtained reactant contains 85.1% of 2,4-dichlorotoluene. If FeCl3 is used as the catalyst to carry out the chlorination reaction at 10~15℃ until the relative density of the solution is 1.025, the product contains 2,4-dichlorotoluene and 3,4-dichlorotoluene, and the mass ratio of the two components is 100:30 . After the chlorination is completed, wash with water to neutrality, and treat with 10% NaOH solution at 100~110℃ to remove other impurities. The treated chloride is rectified and separated in a high-efficiency rectification tower (2,4-dichloro Toluene bp200°C, 3,4-dichlorotoluene bp207°C). The yields of 2,4-dichlorotoluene and 3,4-dichlorotoluene were 64.4% and 19.8%, respectively. Ortho-chlorotoluene method o-chlorotoluene uses sulfuryl chloride as the chlorinating agent to carry out chlorination reaction at 142~196℃. The products are 2,4-dichlorotoluene and 2,3-dichlorotoluene, and unreacted The composition of the raw materials is 55%, 6%, and 39%, respectively. After distillation (2,4-dichlorotoluene b.p. 200°C, 2,3-dichlorotoluene b.p. 207-208°C, o-chlorotoluene b.p. 157-159°C), 2,4-dichlorotoluene was separated. Ortho-nitrotoluene method Ortho-nitrotoluene is chlorinated in the presence of FeCl3 catalyst at 35~40℃. When the relative density of the reactant reaches 1.320 (15℃), wash the material to neutral, and the reactant contains 15% of the raw material , 2-chloro-6-nitrotoluene 49%, 4-chloro-2-nitrotoluene 21%, and 15% polychloride, after rectification and crystallization treatment, the Chemicalbook to 2-chloro-6-nitrotoluene The yields of 4-chloro-2-nitrotoluene and 4-chloro-2-nitrotoluene are 50% and 30% respectively. 4-chloro-2-nitrotoluene is obtained by hydrogenation reduction reaction and steam distillation to obtain 4-chloro-2-amino. Toluene, diazotization and adding CH2Cl2 for Sandmeyer reaction to obtain 2,4-dichlorotoluene. The method is used to produce 4-chloro-2-nitrotoluene which is a by-product of 2-chloro-6-nitrotoluene (used as an intermediate of the herbicide quinclorac). 2,4-Diaminotoluene Method 2,4-Diaminotoluene undergoes diazotization reaction in the presence of NaNO2 and hydrochloric acid, and then performs Sandmeyer reaction in the presence of Cu2Cl2 to obtain 2,4-dichlorotoluene. 3-Chloro-4-methylaniline method 3-chloro-4-methylaniline and hydrochloric acid are added to the reaction kettle, NaNO2 aqueous solution is added dropwise at 3~5℃, and the addition is completed within 2~3h for diazotization After the reaction, the diazotized liquid is added dropwise to a hydrochloric acid solution containing Cu2Cl2 at 2-5°C to perform Sandmeyer reaction to obtain 2,4-dichlorotoluene. Among the above methods, the chloride produced by using p-chlorotoluene and o-chlorotoluene as raw materials contains many impurities and has similar boiling points. It is necessary to use high-efficiency rectifying towers to fractionate to obtain more than 98% of 2,4-dichlorotoluene. These two methods are difficult to operate, and the equipment investment cost is high. The 2,4-diaminotoluene method is not suitable for industrialization, and the o-nitrotoluene method and the 3-chloro-4-methylaniline method to prepare 2,4-dichlorotoluene have the same basic principles, and both require diazotization and Sandmeyer In response, there is a shortcoming of more waste water. The o-nitrotoluene method is used to co-produce 2-chloro-6-nitrotoluene, which is further reduced to obtain 2-chloro-6-aminotoluene, which is an important intermediate for the production of the herbicide quinclorac.
Post time: Apr-28-2021